The strong nuclear force is one of the four fundamental forces of nature and is responsible for holding protons and neutrons together in the nucleus of an atom. It is a very short-lived force that is much stronger than other fundamental forces such as the electromagnetic force. The universe is governed by four fundamental forces that determine the interactions between particles and shape the world as we know it. These forces include electromagnetic force, gravity, weak nuclear force and strong nuclear force. These fundamental forces affect everything in the universe, from the smallest atoms to the largest galaxies.
A recent study by the Argonne National Laboratory and the University of North Carolina at Chapel Hill has brought researchers closer to understanding one of the most mysterious fundamental forces, the strong nuclear force.
His work builds on fundamental theories of atomic structure that emerged in the early 1960s from Argonne physicist and Nobel laureate Maria Heppert Mayer. He helped develop a mathematical model of the core structure. His model explained why a certain number of protons and neutrons in the nucleus of an atom make it extremely stable; this was a phenomenon that had puzzled scientists for some time.
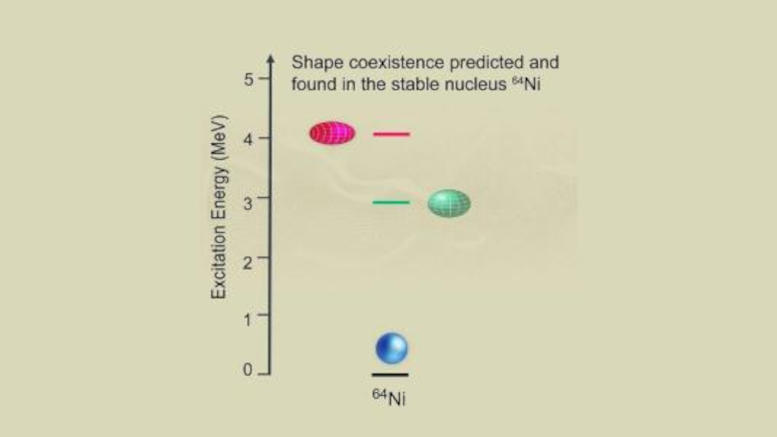
The research team has previously conducted similar experiments to study the strong nuclear force and investigated how the structure of the nucleus can change when it is formed in an excited state by a nuclear reaction. These and other experiments elsewhere led them to study nickel-64, which contains 64 neutrons and protons. This nucleus is the heaviest stable nickel nucleus, with 28 protons and 36 neutrons. This isotope of nickel has properties that allow it to change its structure when excited to higher energy states.
For their experiment, the team used the Argonne Tandem Linac Accelerator System, a DOE Office of Science user facility, to accelerate a sample of Ni-64 cores to a lead target. Lead atoms were able to excite Ni-64 nuclei through electromagnetic forces caused by the repulsion between lead protons and nickel protons.
The process is similar to putting a bag of popcorn in the microwave. As the beans heat up, they begin to take on different shapes and sizes. The popcorn coming out of the microwave oven is different from the one coming in, and more importantly, the kernels have changed shape due to the energy applied to them.
After the Ni-64 nuclei were excited, an instrument called GRETINA detected gamma rays emitted as the nuclei returned to their ground state. Another detector, called CHICO2, determined the direction of the interacting particles. The data obtained by the detectors allowed the team to determine what shape – or shapes – the Ni-64 takes when excited.
From the analysis of the data, it was concluded that the Ni-64 nuclei excited by the interaction with lead also changed shape. But instead of taking the usual puffy shapes, the spherical nickel atomic core morphed into one of two shapes, depending on the amount of energy applied: flattened like a doorknob or elongated like a soccer ball. This finding is unusual for heavy nuclei such as Ni-64, which is composed of many protons and neutrons.
“A model is a representation of reality and is only valid when it can explain what was previously known and has some predictive power,” said Robert Jansens, a UNC-Chapel Hill professor and co-author of the paper. “We study the nature and behavior of nuclei to continually improve our current models of the strong nuclear force.”
Ultimately, the researchers hope that their discoveries in Ni-64 and surrounding nuclei could lay the foundation for future practical discoveries in nuclear science, such as nuclear energy, astrophysics and medicine. “More than 50 percent of medical procedures in hospitals today involve nuclear isotopes,” Jansens said. “And many of these isotopes were discovered while doing basic research like we did.”
Source: Port Altele
